66. Xiong, M.; Chen, F.; Shu, Y.; Wu, X.; Tang, J. Yang, F.*; Xing, D.* Iridium(I)-Catalyzed Atroposelective Alkenylation of Heterobiaryls with Terminal Alkynes. Org. Lett. 2023, 25, 5703–5708

65. Ma, C.; Wang, S.; Sheng, Y.; Zhao, X.; Xing, D.*; Hu, W.* Synthesis and Characterization of Donor-Acceptor Iron Porphyrin Carbenes and Their Reactivities in N–H Insertion and Related Three-Component Reaction. J. Am. Chem. Soc. 2023, 145, 4934–4939.
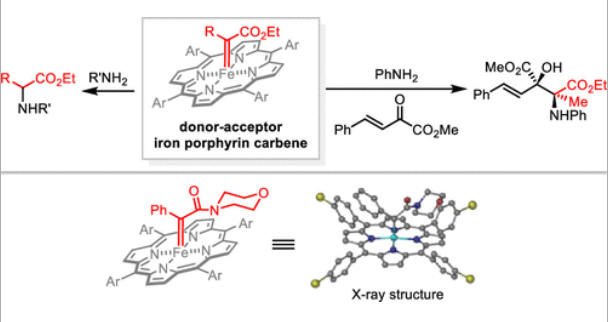

64. Xiang, Y.; Du, R.; Wang, S.; Wu, X.; Tang, J.; Yang, F.*; Xing. D.*KOtBu-catalysed α-homoallylic alkylation of acyclic amides with 1-aryl-1,3-dienes, Mol. Divers 2023, 27, 1481–1487
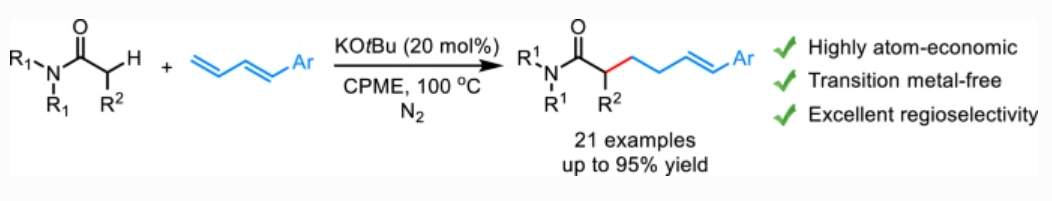
63. Wang, Q.; Yan, Z.; Xing, D.*Nickel(0)-catalysed linear-selective hydroarylation of 2-aminostyrenes with arylboronic acids by a bifunctional temporary directing group strategy, Org. Chem. Front., 2022, 9, 3840–3846.
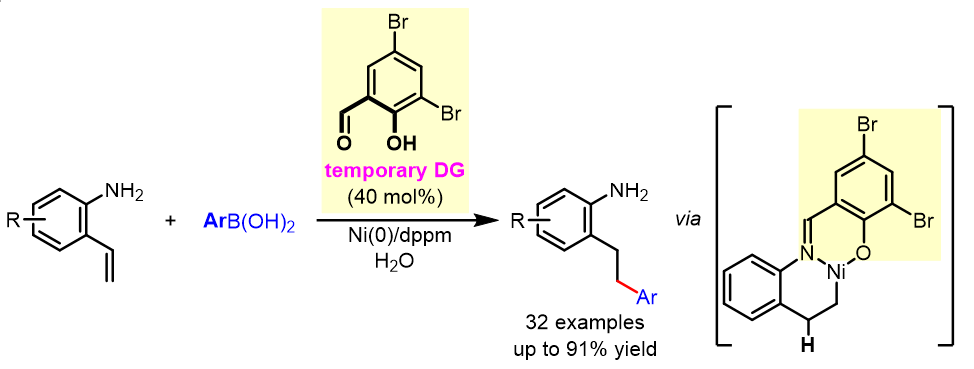
62. Wang, S.; Xiang, Y.; Chen, T.; Wu, X.; Xing, D.*Construction of quaternary carbon centers by KOtBu-catalyzed α-homoallylic alkylation of lactams with 1,3-dienes, Org. Chem. Front., 2022, 9, 1642–1648.
61. Huang, Y.; Zhang, Z.;Xing, D.* Hematite Nanostructure-Based Photoanodes for Interfacial Photoelectrocatalytic Dichlorination of Styrenes, ACS Appl. Nano Mater. 2022, 5, 4609–4614.
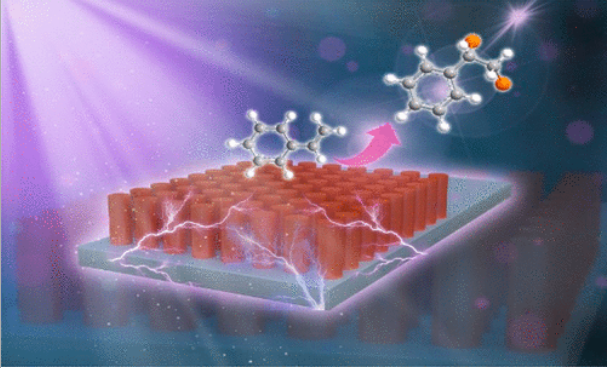
60. Xiong, M.; Shu, Y.; Tang, J.; Yang, F.; Xing, D.*Iridium(I)-Catalyzed Isoindolinone-Directed Branched-Selective Aromatic C–H Alkylation with Simple Alkenes, Molecules, 2022, 27, 1923.

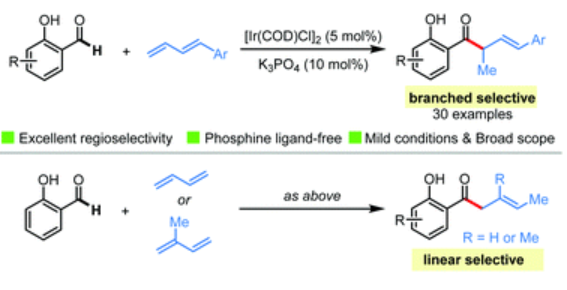
59. Yang, Y.; Xing, D.* Iridium-catalysed branched-selective hydroacylation of 1,3-dienes with salicylaldehydes, Chem. Commun., 2021, 57, 7378–7381.
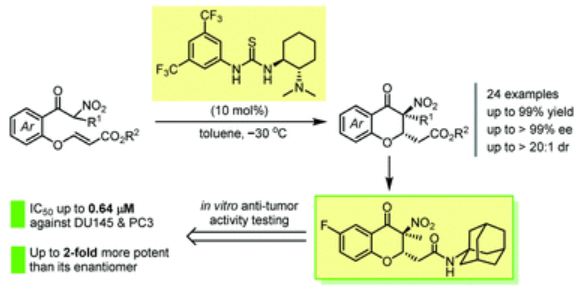
58. Chen, H.; Tang, J.; Liu, T.; Yu, L.-F.; Xing, D.*; Yang, F.* Enantioselective synthesis of chiral 3-alkyl-3- nitro-4-chromanones via chiral thiourea-catalysed intramolecular Michael-type cyclization, Org. Biomol. Chem., 2021, 19, 7403–7408.
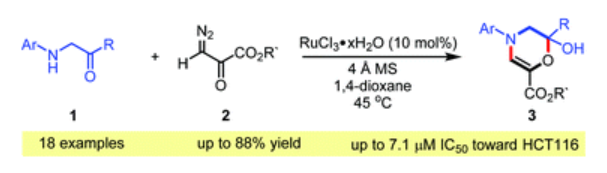
57. Sajjad, F.; Chen, Y.; Tian, X.; Reddy, A. G. K.; Dong, S.; Hu, W.*; Xing, D.* Facile synthesis of 1,4-oxazines by ruthenium-catalyzed tandem N-H insertion/cyclization of α-arylamino ketones and diazo pyruvates. Org. Biomol. Chem., 2021, 19, 1769–1772.
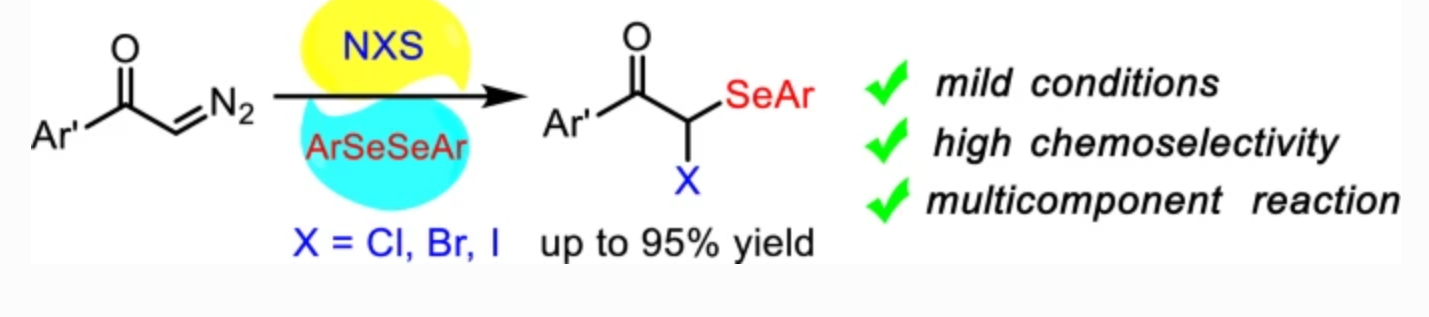
56. Li, J.; Ma, C.; Xing, D.*; Hu, W.* gem-Difunctionalization of α-diazoarylketones with diaryldiselenides and N-halosuccinimides: facile synthesis of α-halo-α-arylseleno ketones. Mol. Divers.2021, 25, 2459–2466.
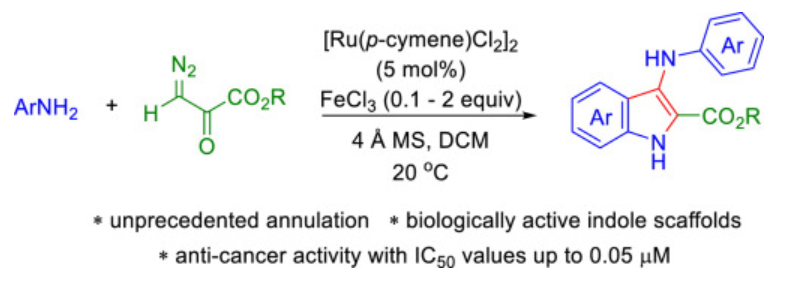
55. Sajjad, F.; Reddy, A. G. K.; Xing, D.*; Dong, S.; Hu, W., Ruthenium(II)-catalyzed facile synthesis of 3-(phenylamino)-1H-indole-2-carboxylates from anilines and diazo pyruvates promoted by FeCl3. Tetrahedron. 2021, 77, 131399.
54. Chen, T.; Yang, H.; Yang, Y.; Dong, G.*; Xing, D.* Water-Accelerated Nickel-Catalyzed α-Crotylation of Simple Ketones with 1,3-Butadiene under pH and Redox-Neutral Conditions, ACS Catal. 2020, 10, 4238–4243.

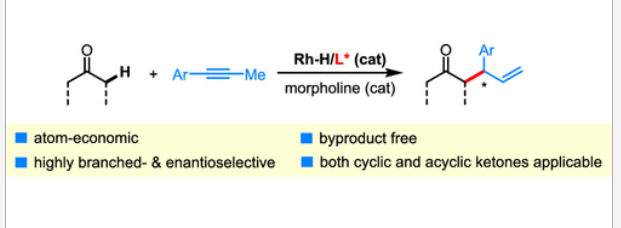
53. Xie, L.; Yang, H.; Ma, M.; Xing, D.* Rhodium-Catalyzed Branched and Enantioselective Direct α-Allylic Alkylation of Simple Ketones with Alkynes. Org. Lett. 2020, 22, 2007–2011.
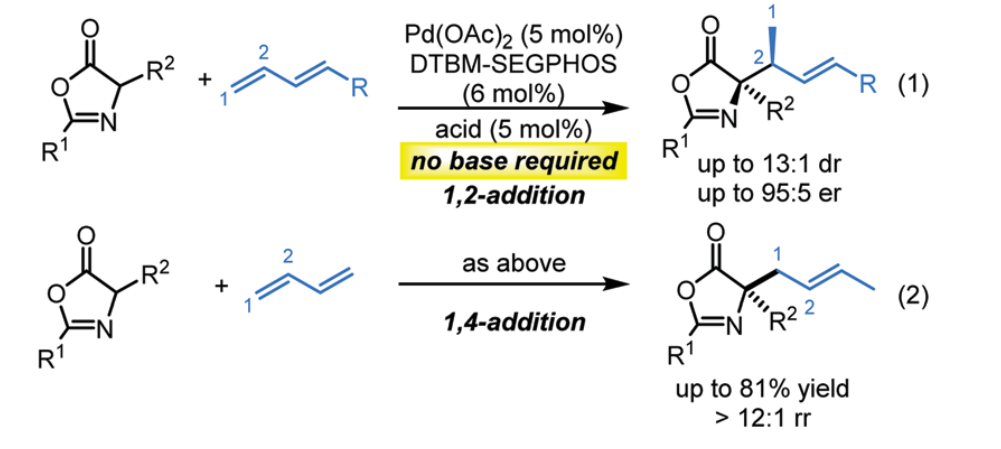
52. Yang, H.; Xing, D.* Palladium-catalyzed diastereo- and enantioselective allylic alkylation of oxazolones with 1,3-dienes under base-free conditions, Chem. Commun. 2020, 56, 3721–3724.

51. Che, J.; Niu, L.; Jia, S.; Xing, D.*; Hu, W.* Enantioselective Three-Component Aminomethylation of α-Diazo Ketones with Alcohols and 1,3,5-Triazines. Nat. Commun. 2020, 11, 1511.
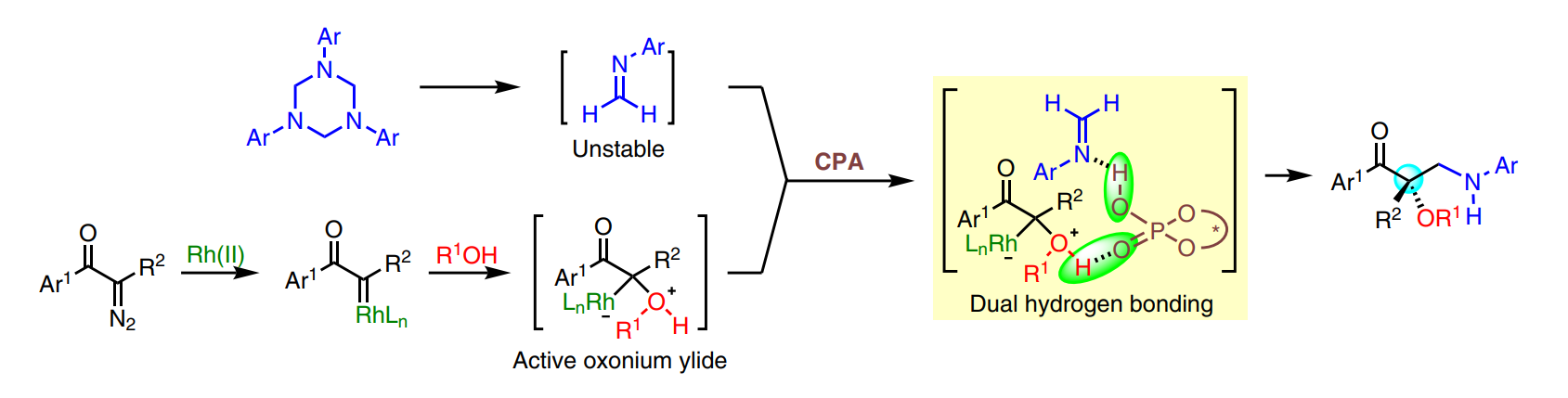
50. Sajjad, F.; Reddy, A. G. K.; Che, J.; Hu, W.*; Xing, D.* Ruthenium-Catalyzed Diastereoselective Synthesis of Fully Substituted Pyrrolidines from Anilines and Diazo Pyruvates. Org. Lett. 2020, 22, 3094-3098.
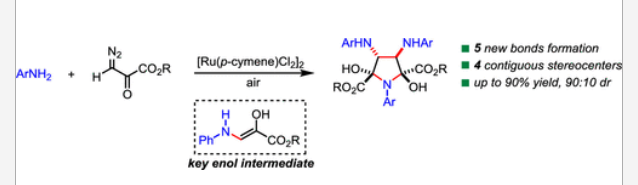
49. Lin, Y.; Xing, D.; Wu, W.-B.; Xu, G.-Y.; Yu, L.-F.; Tang, J.; Zhou, Y.-B.; Li, J.; Yang, F.*, Design, synthesis, and in vitro evaluation of benzofuro[3,2-c]quinoline derivatives as potential antileukemia agents. Molecules2020, 25, 203.
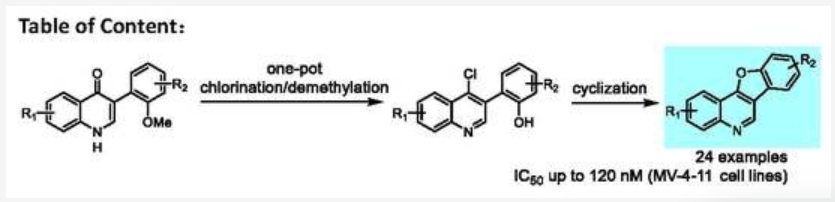
48. Kang, Z.; Shou, J.; Xing, D.; Hu, W.*, Rh(II)/Ag(I)-Cocatalyzed Three-Component Reaction via SN1/SN1'-Type Trapping of Oxonium Ylide with the Nicholas Intermediate. J. Org. Chem.2020, 85, 9850–9862.
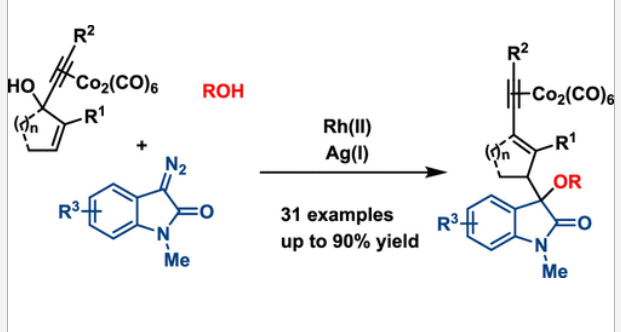
47. Ji, J.; Sajjad, F.; You, Q.; Xing, D.; Fan, H.; Reddy, A. G. K.; Hu, W.*; Dong, S.*, Synthesis and biological evaluation of substituted pyrrolidines and pyrroles as potential anticancer agents. Arch. Pharm.2020, 353, 2000136.
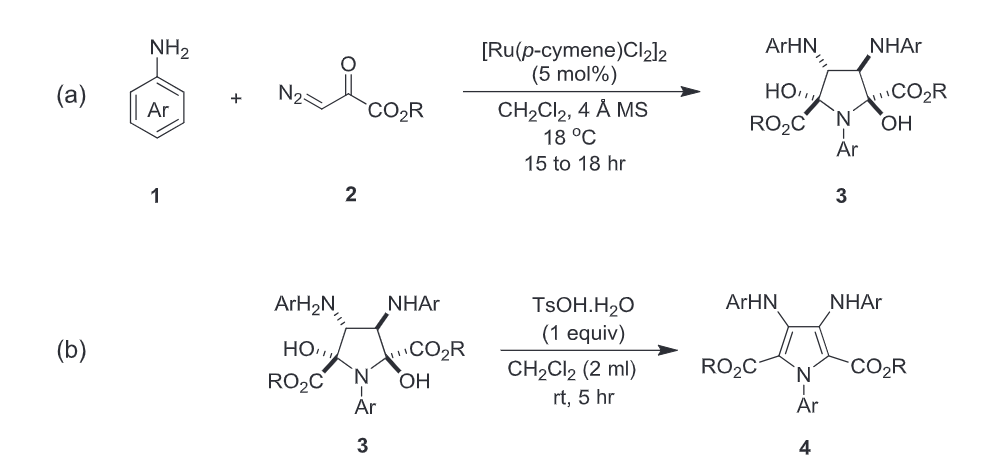
46. Che, J.; Gopi Krishna Reddy, A.; Niu, L.; Xing, D.*; Hu, W.* Cu(I)-Catalyzed Three-Component Reaction of α-Diazo Amide with Terminal Alkyne and Isatin Ketimine via Electrophilic Trapping of Active Alkynoate-Copper Intermediate. Org. Lett. 2019, 21, 4571–4574.
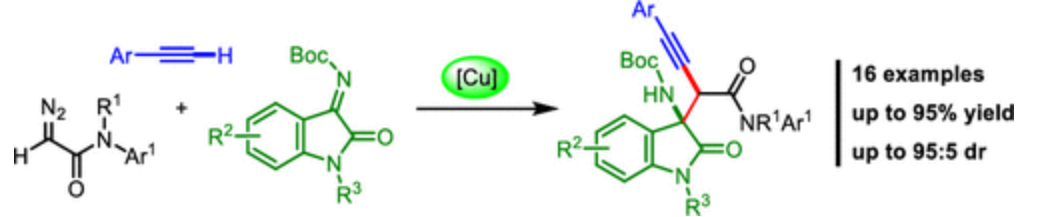
45. Li, J.; Ma C.; Xing D.*; Hu W* Catalyst-Free gem-Difunctionalization of Fluoroalkyl-Substituted Diazo Compound with Diselenide or Disulfide and NFSI. Org. Lett. 2019, 21, 2101–2105.
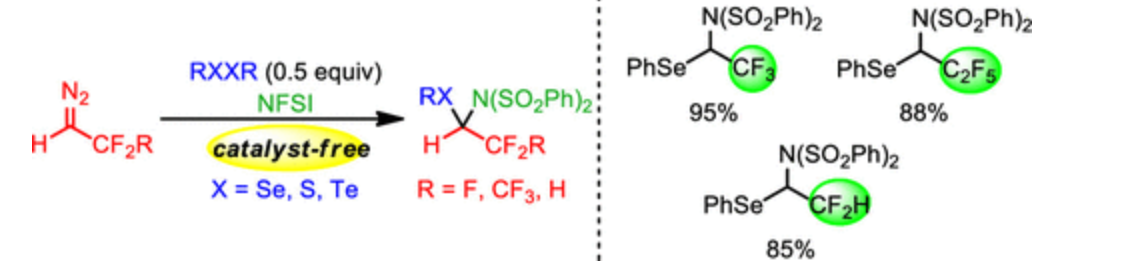
44. Xing, D.; Qi X.; Marchant D.; Liu P.*; Dong G.* Branched-Selective Direct -Alkylation of Cyclic Ketones with Simple Alkenes. Angew. Chem. Int. Ed. 2019, 58 (13), 4366–4370.
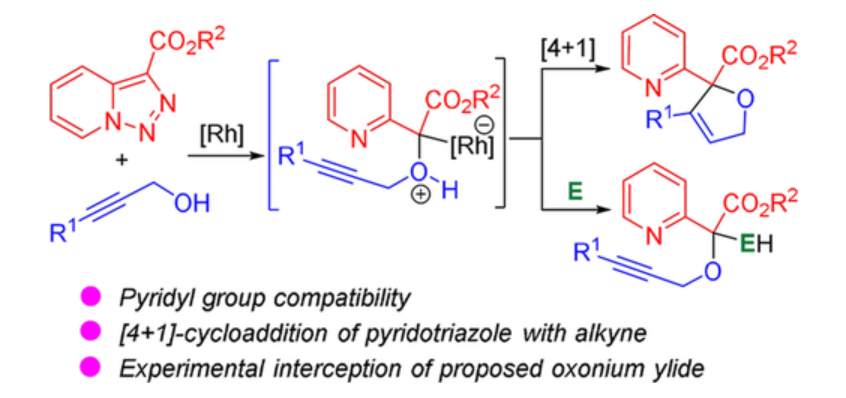
43. Lv, X.; Yang, H.; Shi, T.; Xing, D.; Xu, X.*; Hu, W.*, Rhodium(II)-Catalyzed Formal [4+1]-Cycloaddition of Pyridotriazoles and Propargyl Alcohols: Synthesis of 2,5-Dihydrofurans. Adv. Synth. Catal. 2019,361, 1265–1270.
42. Lim, H. N.; Xing, D.; Dong, G.*, Transition-Metal-Catalyzed Ketone α-Alkylation and Alkenylation with Simple Alkenes and Alkynes through a Dual Activation Strategy. Synlett 2019,30, 674-684.

41. Chen, H.; Xing, Y.; Xie, J.; Xie, J.; Xing, D.; Tang, J.; Yang, F.; Yi, Z.*; Qiu, W.-W.*, Synthesis and biological evaluation of 3-nitro-4-chromanone derivatives as potential antiproliferative agents for castration-resistant prostate cancer. RSC Adv. 2019,9, 33794–33799.
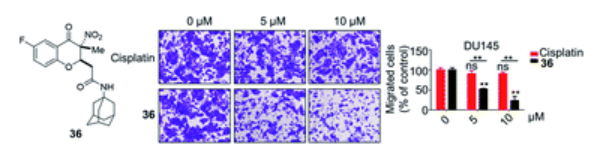
40. Chen, H.; Xie, J.; Xing, D.; Wang, J.; Tang, J.; Yi, Z.; Xia, F.; Qiu, W.-W.*; Yang, F.*, Diastereoselective synthesis of 3,3-disubstituted 3-nitro-4-chromanone derivatives as potential antitumor agents. Org. Biomol. Chem. 2019,17, 1062–1066.

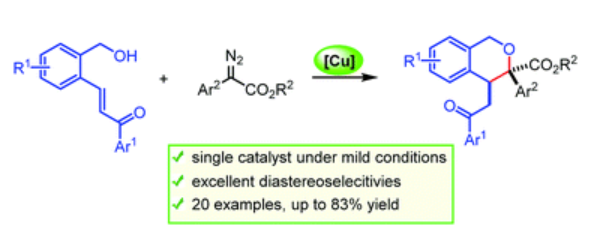
39. Alavala, G. K. R.; Sajjad, F.; Shi, T.; Kang, Z.; Ma, M.; Xing, D.*; Hu, W.* Diastereoselective synthesis of isochromans via the Cu(II)-catalysed intramolecular Michael-type trapping of oxonium ylides. Chem. Commun. 2018, 54, 12650–12653.
38. Lv, X.; Kang, Z.; Xing, D.*; Hu, W.* Cu(I)-Catalyzed Three-Component Reaction of Diazo Compound with Terminal Alkyne and Nitrosobenzene for the Synthesis of Trifluoromethyl Dihydroisoxazoles. Org. Lett. 2018, 20, 4843–4847.

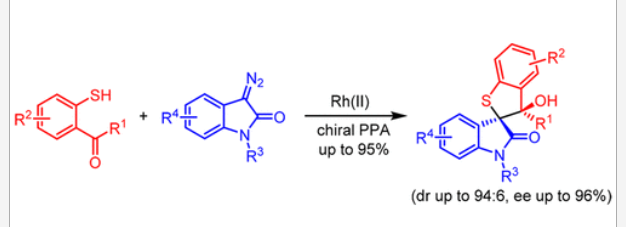
37. Xiao, G.; Chen, T.; Ma, C.; Xing, D.*; Hu, W.* Rh(II)/Chiral Phosphoric Acid-Cocatalyzed Enantioselective Synthesis of Spirooxindole-Fused Thiaindans. Org. Lett. 2018, 20, 4531–4535.
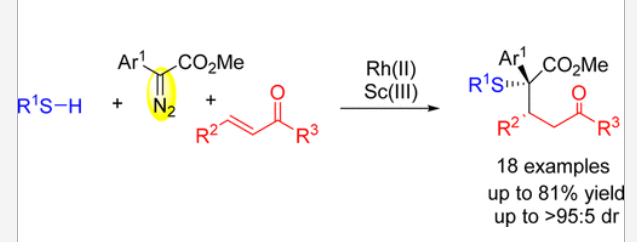
36. Xiao, G.; Ma, C.; Wu, X.; Xing, D.*; Hu, W.* Synthesis of γ-Sulfur-Substituted Ketones via Rh(II)/Sc(III) Cocatalyzed Three-Component Reaction of Diazo Compounds with Thiophenols and Enones. J. Org. Chem. 2018, 83, 4786–4791.
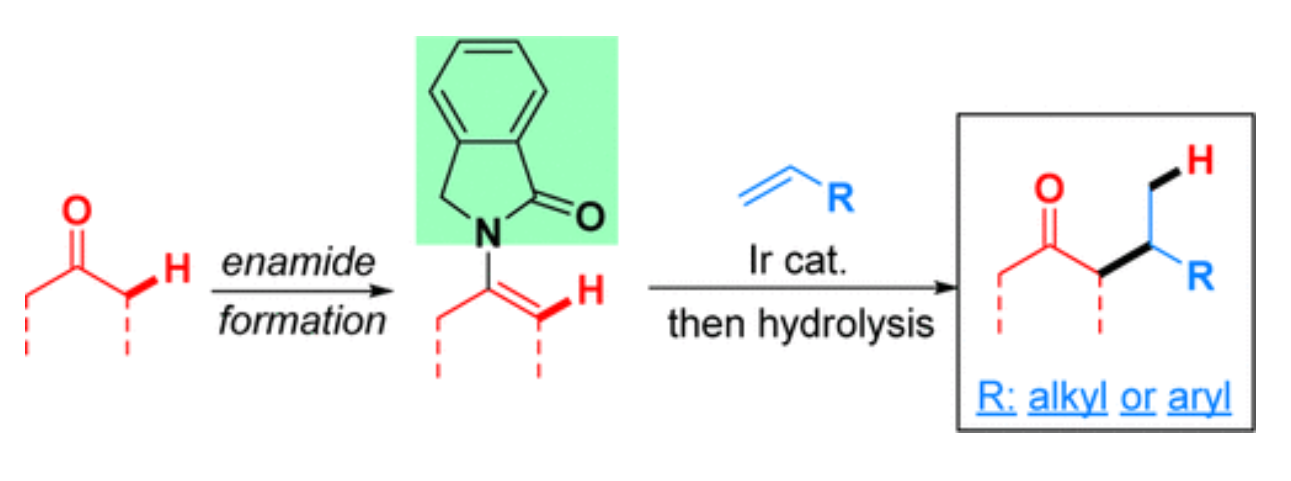
35. Xing, D.*; Dong, G.* Branched-Selective Intermolecular Ketone -Alkylation with Unactivated Alkenes via An Enamide Directing Strategy. J. Am. Chem. Soc. 2017, 139, 13664–13667.
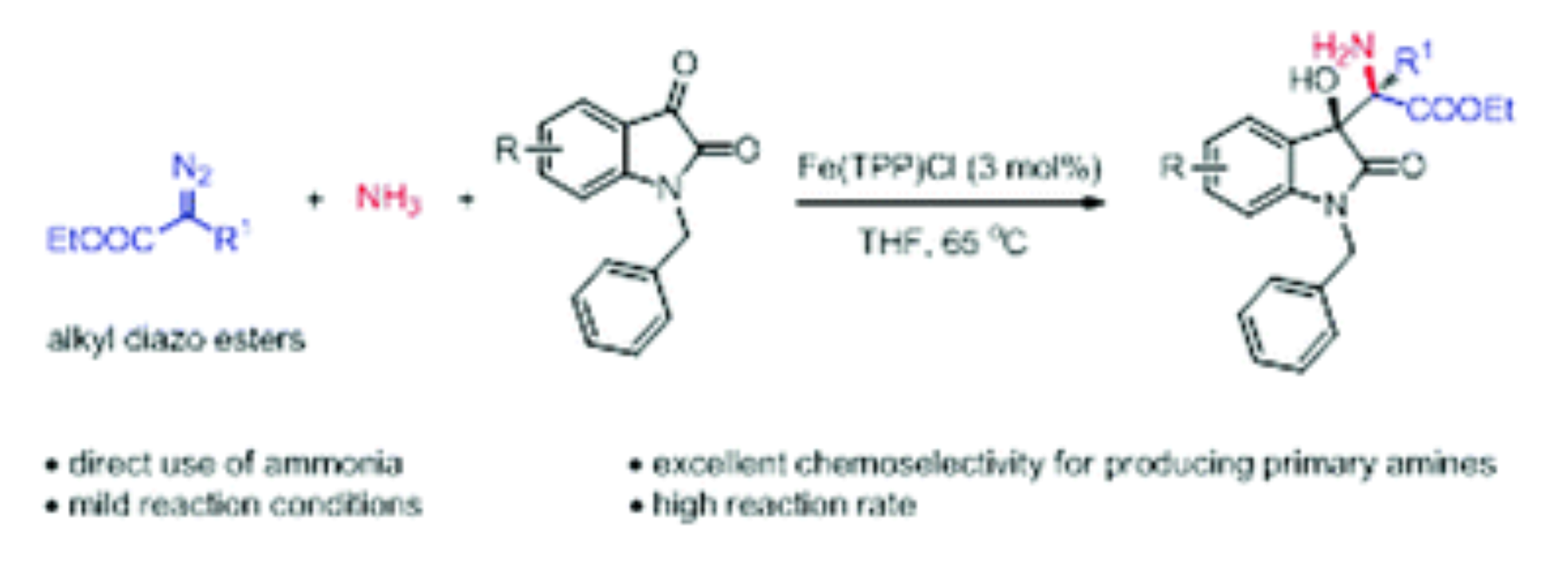
34. Ma, C.; Chen, J.; Xing, D.*; Sheng, Y.; Hu, W.*, Iron catalyzed efficient synthesis of poly-functional primary amines via the direct use of ammonia. Chem. Commun.2017, 53, 2854–2857.
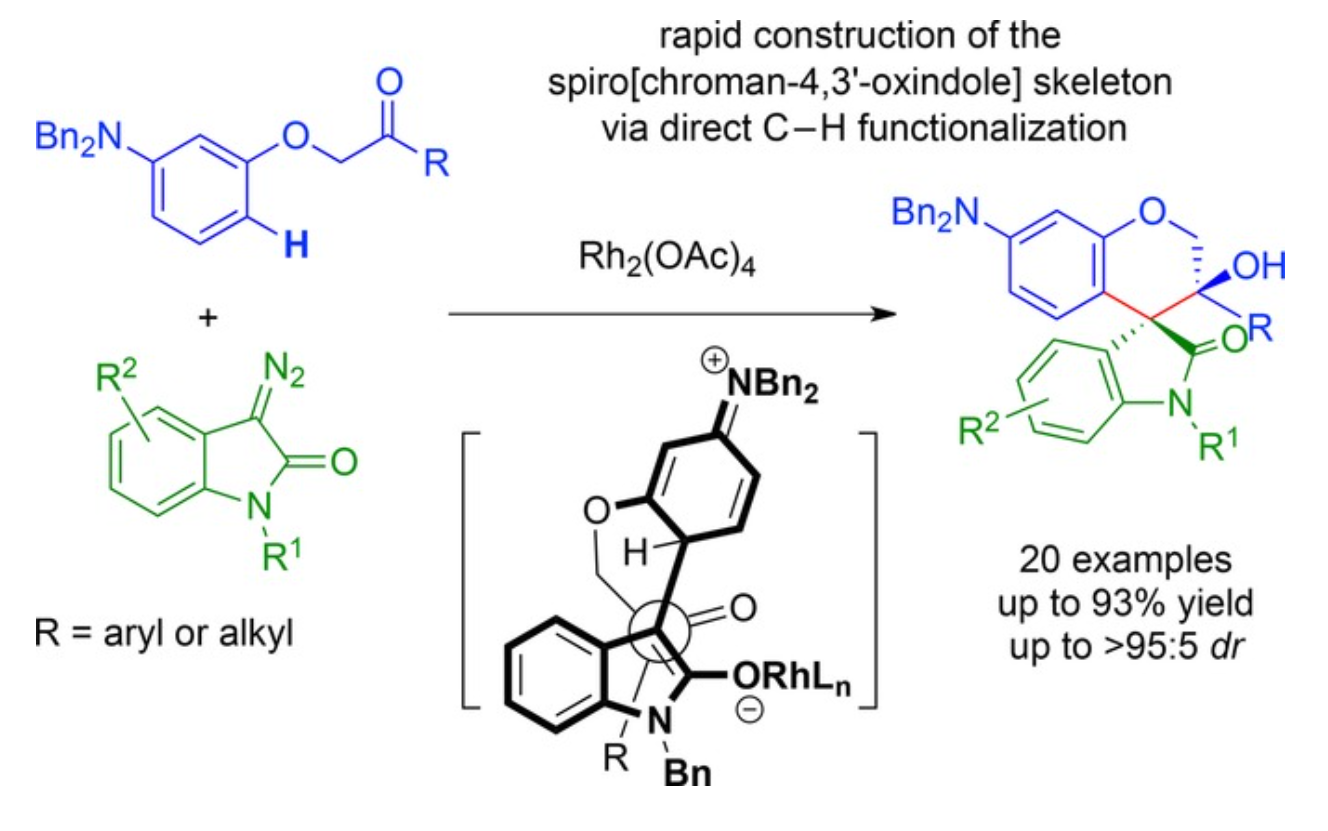
33. Jia, S.; Lei, Y.; Song, L.; Krishna Reddy, A. G.; Xing, D.; Hu, W.*, Diastereoselective Intramolecular Aldol-Type Trapping of Zwitterionic Intermediates by Ketones for the Synthesis of Spiro[chroman-4,3'-oxindole] Derivatives. Adv. Synth. Catal.2017, 359, 58–63.
32. Jia, K.; Lv, X.; Xing, D.; Che, J.; Liu, D.; Thumar, N. J.; Dong, S.*; Hu, W.*, Synthesis and biological evaluation of 3-amino-3-hydroxymethyloxindoles as potential anti-cancer agents. RSC Adv.2017, 7, 23265–23271.
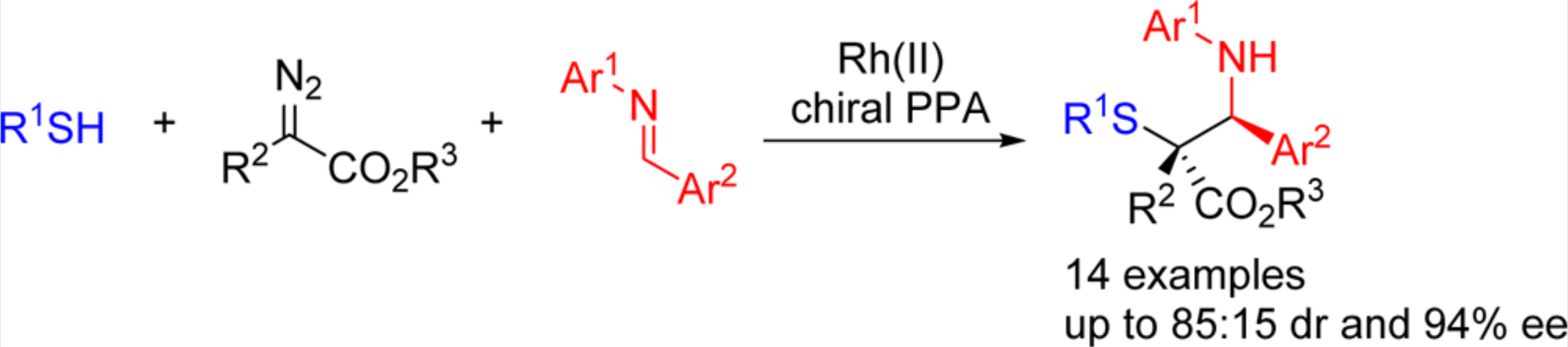
31. Xiao, G.; Ma, C.; Xing, D.*; Hu, W.* Enantioselective Synthesis of α-Mercapto-β-amino Esters via Rh(II)/Chiral Phosphoric Acid-Cocatalyzed Three-Component Reaction of Diazo Compounds, Thiols, and Imines. Org. Lett. 2016, 18, 6086–6089
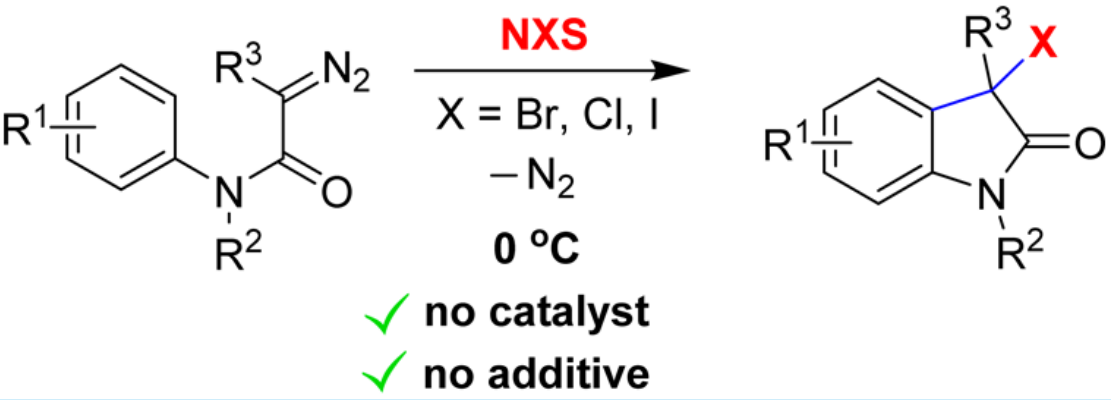
30. Ma, C.; Xing, D.*; Hu, W.* Catalyst-Free Halogenation of α-Diazocarbonyl Compounds with N-Halosuccinimides: Synthesis of 3-Halooxindoles or Vinyl Halides. Org. Lett. 2016, 18, 3134–3137
29. Che, J.; Xing, D.*; Hu, W.* Metal-Catalyzed Cross-Coupling of Terminal Alkynes with Different Carbene Precursors. Curr. Org. Chem. 2016, 20, 41–60
28. Jia, S.-K.; Song, L.-L.; Lei, Y.-B.; Gopi Krishna Reddy, A.; Xing, D.*; Hu, W.-H.*, A Rh(II)-catalyzed three-component reaction of 3-diazooxindoles with N,N-disubstituted anilines and glyoxylates for the synthesis of 3-aryl-3-substituted oxindoles. Org. Biomol. Chem.2016, 14, 10157–10160.
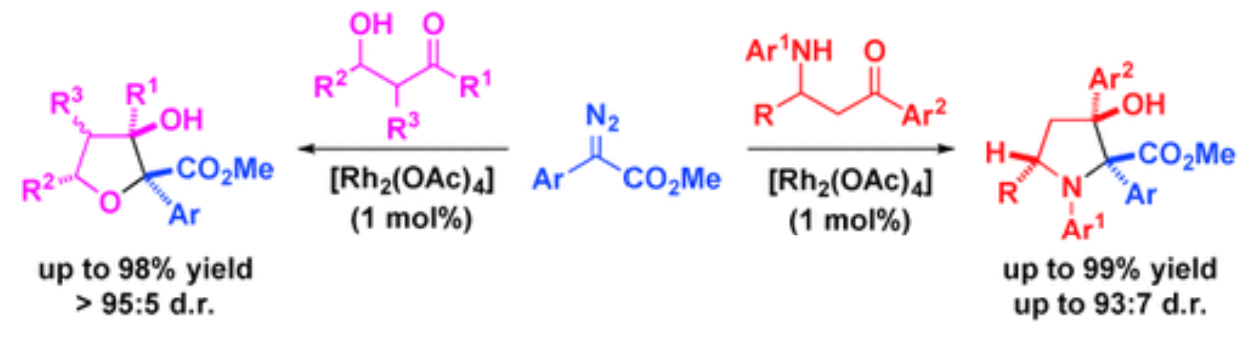
27. Jing, C.; Xing, D.*; Gao, L.; Li, J.; Hu, W.* Divergent Synthesis of Multisubstituted Tetrahydrofurans and Pyrrolidines via Intramolecular Aldol-type Trapping of Onium Ylide Intermediates, Chem. Eur. J.2015, 21, 19202–19207
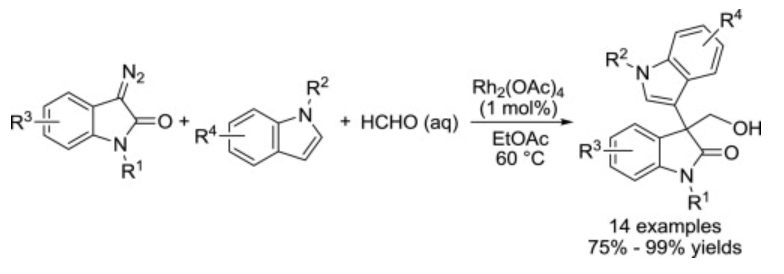
26. Jing, C.; Xing, D.*; Hu, W.* Catalytic Asymmetric Four-Component Reaction for the Rapid Construction of 3,3-Disubstituted 3 Indol-3’-yloxindoles. Org. Lett. 2015,17, 4336–4339.
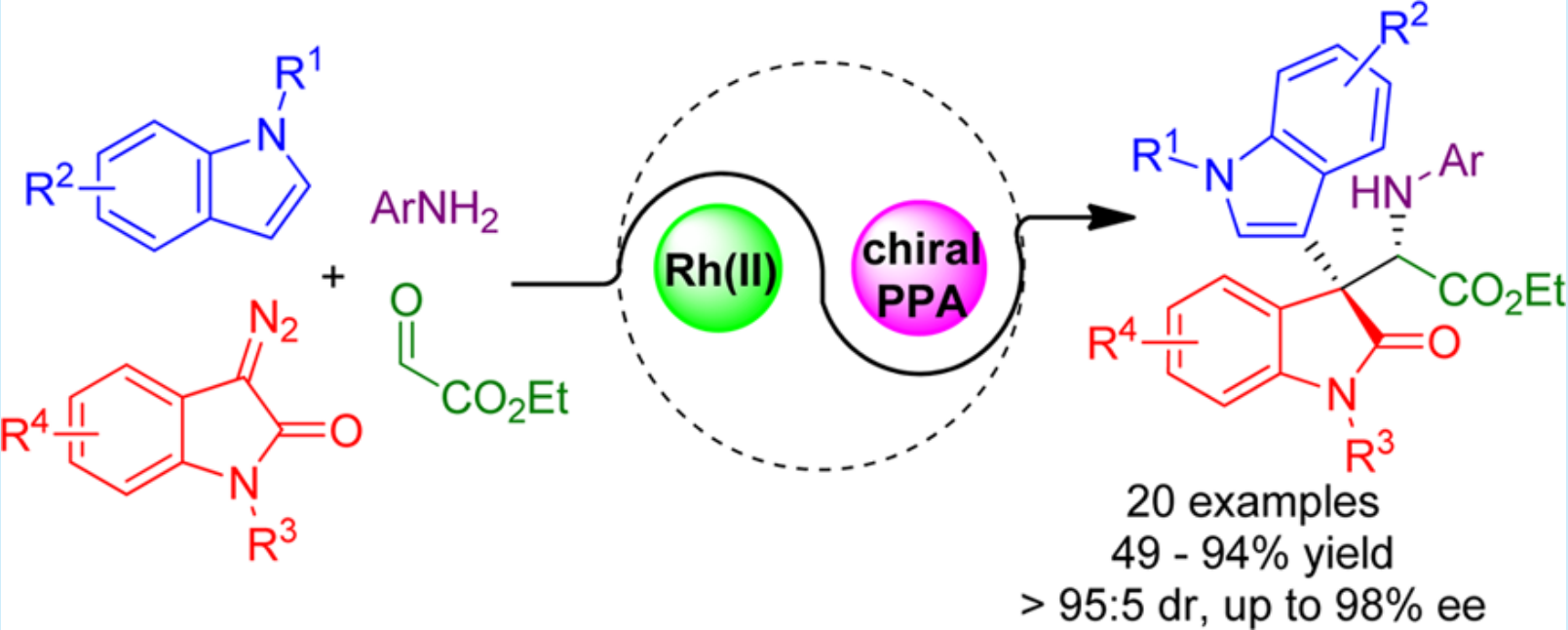
25. Tang, M.; Xing, D.*; Huang, H.; Hu, W.* Divergent synthesis of chiral heterocycles via sequencing of enantioselective three-component reactions and one-pot subsequent cyclization reactions. Chem. Commun. 2015,51, 10612–10615. (Inside front cover)


24. Jing, C.; Xing, D.*; Hu, W.* Synthesis of 3-(hydroxymethyl)-3-indol-3`-yloxindoles via Rh(II)-catalyzed three-component reaction of 3-diazooxindoles, indoles and formalin. Tetrahedron2015, 71, 3597–3602.
23. Tang, M.; Xing, D.*; Hu, W.* Diazo Compounds-Involved Catalytic Asymmetric Multicomponent Reactions, Chin. J. Org. Chem.2014, 34, 1268–1276.
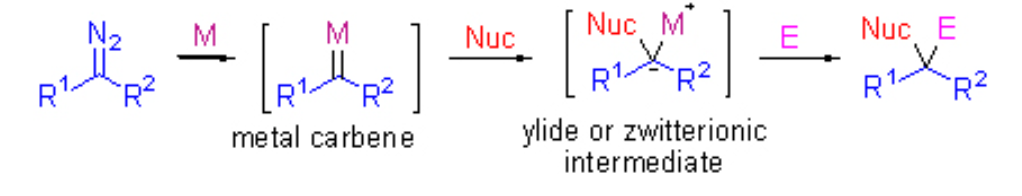
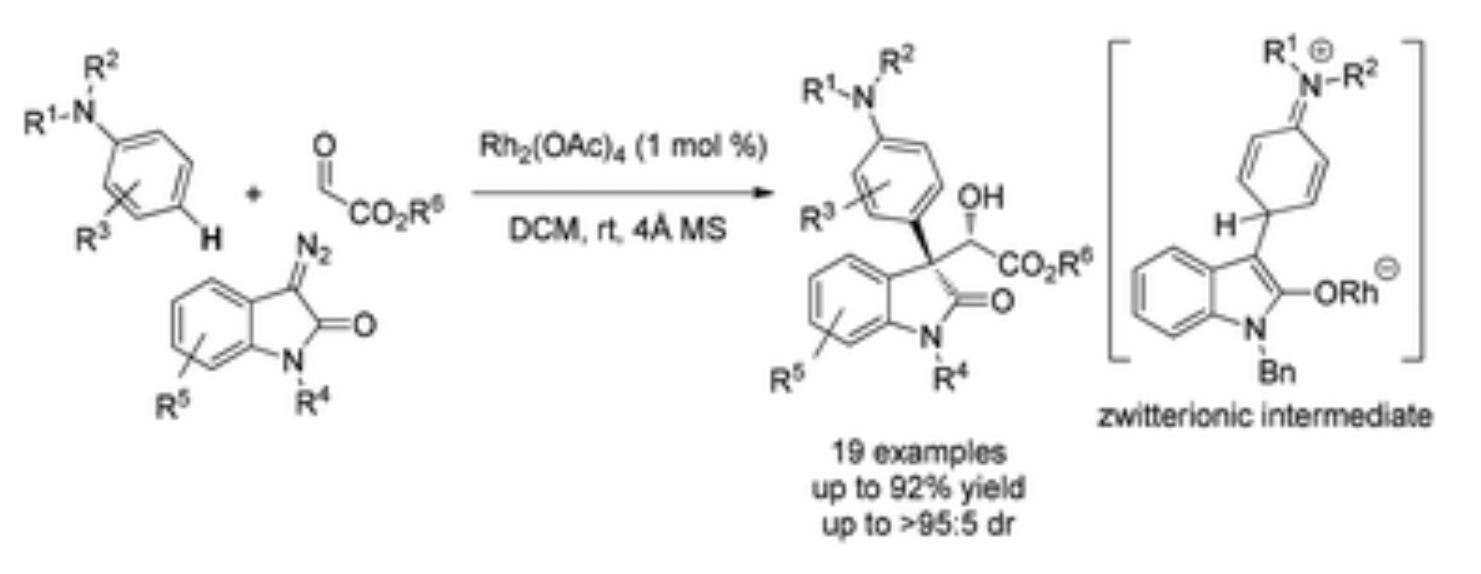
22. Jia, S.; Xing, D.*; Zhang, D; Hu, W.* Catalytic Asymmetric Aromatic C–H Functionalization via Electrophilic Trapping of Active Zwitterionic Intermediate. Angew. Chem. Int. Ed.2014,53 (48), 13098–13101. (Inside Cover; Highlighted by Synfacts: 2014, 10, 1171
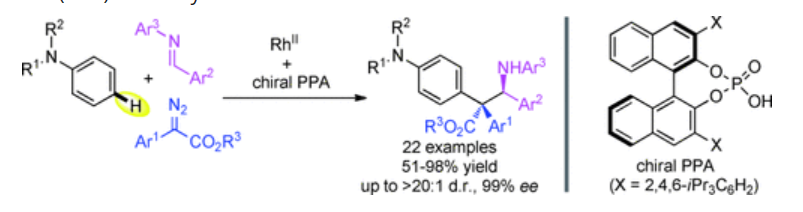

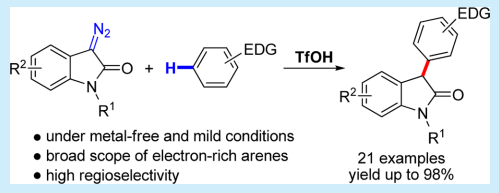
21. Zhai, C.; Xing, D.*; Jing, C.; Zhou, J.; Wang, C.; Wang, D.; Hu, W.* Facile Synthesis of 3-Aryloxindoles via Brønsted Acid Catalyzed Friedel–Crafts Alkylation of Electron-Rich Arenes with 3-Diazooxindoles. Org. Lett. 2014,16, 2934–2937.
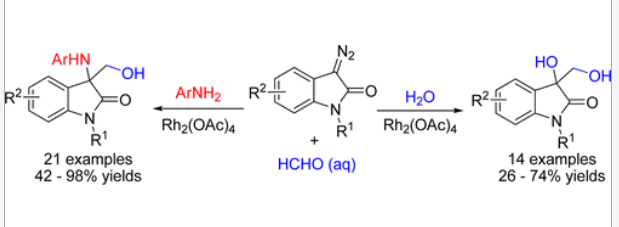
20. Wang, C.; Xing, D.*; Wang, D.; Wu, X.; Hu, W.* Synthesis of 3-Amino-3-hydroxymethyloxindoles and 3-Hydroxy-3-hydroxymethyloxindoles by Rh2(OAc)4-Catalyzed Three-Component Reactions of 3-Diazooxindoles with Formaldehyde and Anilines or Water. J. Org. Chem.2014, 79, 3908–3916.
19. Zhai, C.; Xing, D.*; Qian, Y.; Ji, J.; Ma, C.; Hu, W.* Trapping of Carboxylic Oxonium Ylides with N-Boc Imines for the Facile Synthesis of β-Amino Alcohol Derivatives. Synlett2014, 25, 1745–1750.

18. Yang, S.; Shi, W.; Xing, D.; Zhao, Z.*; Lv, F.; Yang, L.; Yang, Y.*; Hu, W.*, Synthesis, antibacterial activity, and biological evaluation of formyl hydroxyamino derivatives as novel potent peptide deformylase inhibitors against drug-resistant bacteria. Eur. J. Med. Chem. 2014,86, 133–152.

17. Xing, D.; Hu, W.*, Recent advances in metal carbenoid mediated nitrogen-containing zwitterionic intermediate trapping process. Tetrahedron Lett. 2014,55, 777–783.
16. Jing, C.; Xing, D.*; Qian, Y.; Hu, W.*, Rh2(OAc)4 and chiral phosphoric acid cocatalyzed highly diastereo- and enantio-selective four-component reactions: facile synthesis of chiral α,β-diamino acid derivatives. Synthesis 2014,46, 1348.

15. Jing, C.; Xing, D.*; Hu, W.*, Highly diastereoselective synthesis of 3-hydroxy-2,2,3-trisubstituted indolines via intramolecular trapping of ammonium ylides with ketones. Chem. Commun. 2014,50, 951–953.
14. Xing, D.*; Jing, C.; Li, X.; Qiu, H.; Hu, W.* Highly Efficient Synthesis of Mixed 3,3′-Bisindoles via Rh(II)-Catalyzed Three-Component Reaction of 3-Diazooxindoles with Indoles and Ethyl Glyoxylate. Org. Lett.2013, 15, 3578–3581. (Highlighted by Synfacts: 2013, 9, 1078)
13. Ma, C.; Xing, D.*; Zhai, C.; Che, J.; Liu, S.; Wang, J.; Hu, W.* Iron Porphyrin-Catalyzed Three-Component Reaction of Ethyl Diazoacetate with Aliphatic Amines and β,γ-Unsaturated α-Keto Esters. Org. Lett. 2013, 15, 6140–6143.
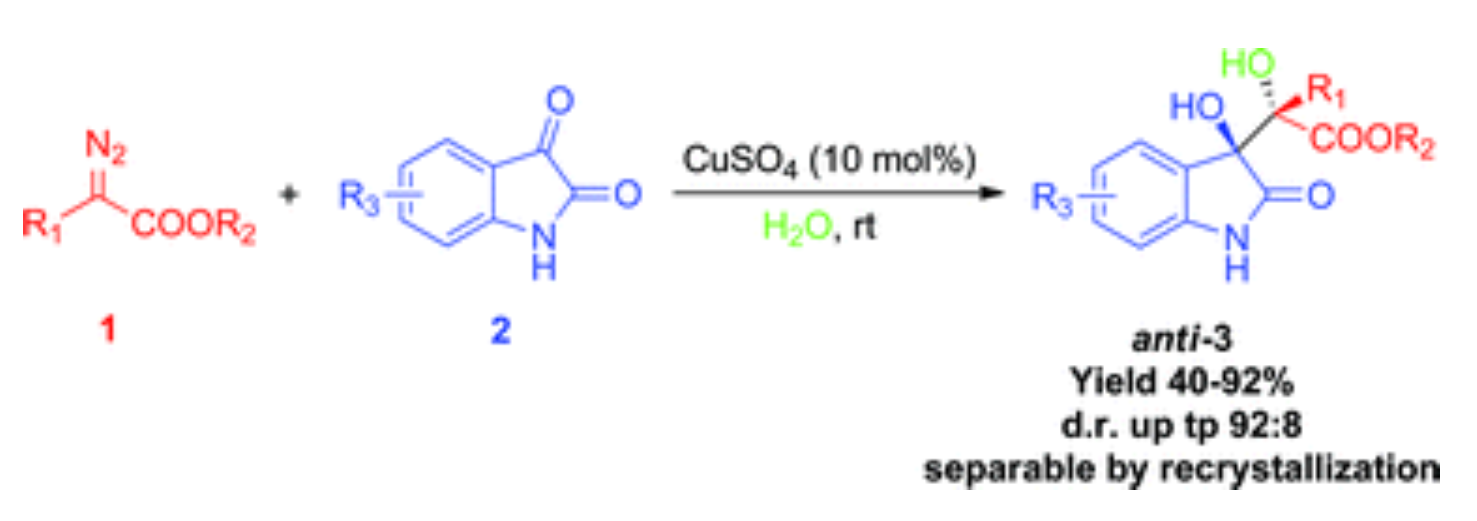
12. Jing, C.; Shi, T.; Xing, D.; Guo, X.; Hu, W.* CuSO4-catalyzed three-component reaction of α-diazo ester, water and isatin: an efficient approach to oxindole derivatives, Green Chem.2013, 15, 620–624.
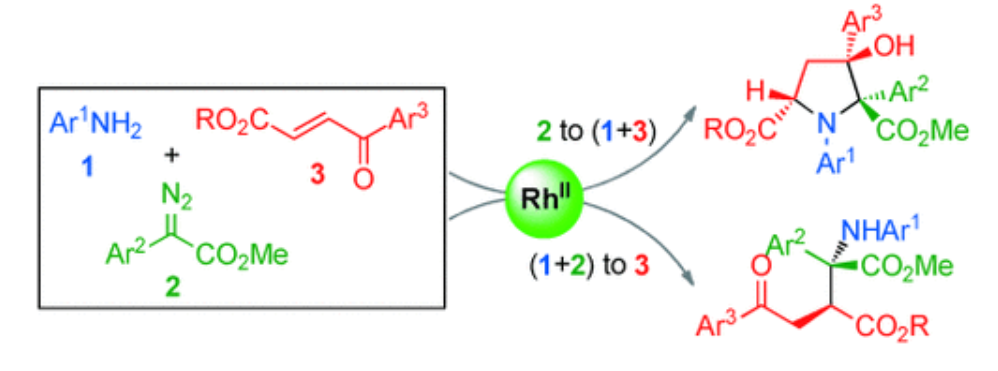
11. Jing, C.; Xing, D.; Qian, Y.; Shi, T.; Zhao, Y.; Hu, W.* Diversity-Oriented Three-Component Reactions of Diazo Compounds with Anilines and 4-Oxo-Enoates. Angew. Chem. Int. Ed.2013, 52, 9289–9292.
10. Wang, C.; Liu, S.; Xing, D.; Wang, X.; Wu, X.; Hu, W.*, Efficient synthesis of α-aryl serine derivatives via three-component reactions of aryldiazoacetates, anilines and formaldehyde. Tetrahedron2013, 69, 11203-11208.
Book Chapter:
Xing, D. and Hu, W. (2014), “Multicomponent Reactions via Active Intermediate of Metal Carbenoid” Chap. 7 in Multicomponent Reactions in Organic Synthesis, Zhu, J. Ed., Wiley-VCH: Weinheim.
Prior ECNU:
9. Xing, D.; Yang, D.* Pd(II)-Catalyzed Intramolecular 1,2-Aminoalkylation of Conjugated 1,3-Dienes for the Synthesis of Pyrrolizidines. Org. Lett. 2013,15, 4370-4373.
8. Xing, D.; Yang, D. Gold(I)-Catalyzed Decarboxylative Allylic Amination of Allylic N-Tosylcarbamates, Org. Synth. 2012, 89, 450–459
7. Xing, D.; Yang, D.* Highly efficient gold(I)-catalyzed Overman rearrangement in water. Beilstein J. Org. Chem.2011, 7, 781–785
6.Xing, D.; Yang, D.* Gold(I)-Catalyzed Highly Regio- and Stereoselective Decarboxylative Amination of Allylic N-Tosylcarbamates via Base-Induced Aza-Claisen Rearrangement in Water. Org. Lett. 2010, 12, 1068–1071.
5. Xing, D.; Xu, X.; Yang, L.*, Highly chemoselective rearrangement of 3-aryloxaziridines to nitrones or amides. Synthesis2009, 20, 3399-3404.
4. Li, H.; Guan, B.; Wang, W.; Xing, D.; Fang, Z.; Wan, X.; Yang, L.; Shi, Z.*, Aerobic oxidation of alcohol in aqueous solution catalyzed by gold. Tetrahedron2007, 63, 8430-8434.
3. Xing, D.; Guan, B.; Cai, G.; Fang, Z.; Yang, L.; Shi, Z.*, Gold(I)-Catalyzed Oxidative Cleavage of a C-C Double Bond in Water. Org. Lett.2006, 8, 693-696.
2. Wan, X.; Xing, D.; Fang, Z.; Li, B.; Zhao, F.; Zhang, K.; Yang, L.; Shi, Z.*, Multiple Deprotonations and Deaminations of Phenethylamines to Synthesize Pyrroles. J. Am. Chem. Soc.2006, 128, 12046-12047.
1. Guan, B.; Xing, D.; Cai, G.; Wan, X.; Yu, N.; Fang, Z.; Yang, L.; Shi, Z.*, Highly Selective Aerobic Oxidation of Alcohol Catalyzed by a Gold(I) Complex with an Anionic Ligand. J. Am. Chem. Soc.2005, 127, 18004-18005.
